THEORY
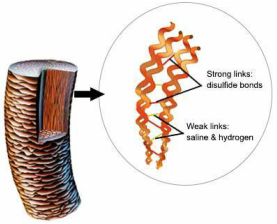 Diagram illustrating bonds in hair strand
Diagram illustrating bonds in hair strand
Hair consists of a protein known as keratin. Keratin is known to give hair its strength and elasticity. Keratin is made up of numerous amino acids that contain sulfur. When these sulfurs atoms bond together they create disulphide bonds in the hair which is what makes hair strong and retain a specific shape. To be able to achieve straightening of the hair shaft, the bonds that hold hair together must be broken.
Straightening techniques such as using a flat iron are temporary as they only use heat. The excessive heat breaks the disulphide bonds temporarily. This temporary breakages allows the strands to be rearranged in a straight position and as the hair cools the bonds are reformed. However humidity or getting the hair wet reverts it the hair to its original form.
Straightening techniques such as using a flat iron are temporary as they only use heat. The excessive heat breaks the disulphide bonds temporarily. This temporary breakages allows the strands to be rearranged in a straight position and as the hair cools the bonds are reformed. However humidity or getting the hair wet reverts it the hair to its original form.
 Reformation of disulphide bond after straightening
Reformation of disulphide bond after straightening
When trying to achieve permanent straightening a strong reducing agent is used to break the disulphide bonds. A strong alkaline solution such as sodium hydroxide or ammonium thioglycolate are used for this first step. Once applied and left to sit for a while the hair is combed straight and held in that position while acidic solutions are added. The acid neutralizes the base thereby allowing the bonds to form in their new positions.
Alkaline hydroxides are the harshest chemicals to use for this process and there is danger in leaving them in the hair for too long. However they work far more quickly than the thioglycolate solutions. Thioglycolate solutions are much safer for the hair and if used, a treatment of an oxidizing agent such as hydrogen peroxide is necessary to allow the new bonds to form.
Alkaline hydroxides are the harshest chemicals to use for this process and there is danger in leaving them in the hair for too long. However they work far more quickly than the thioglycolate solutions. Thioglycolate solutions are much safer for the hair and if used, a treatment of an oxidizing agent such as hydrogen peroxide is necessary to allow the new bonds to form.
